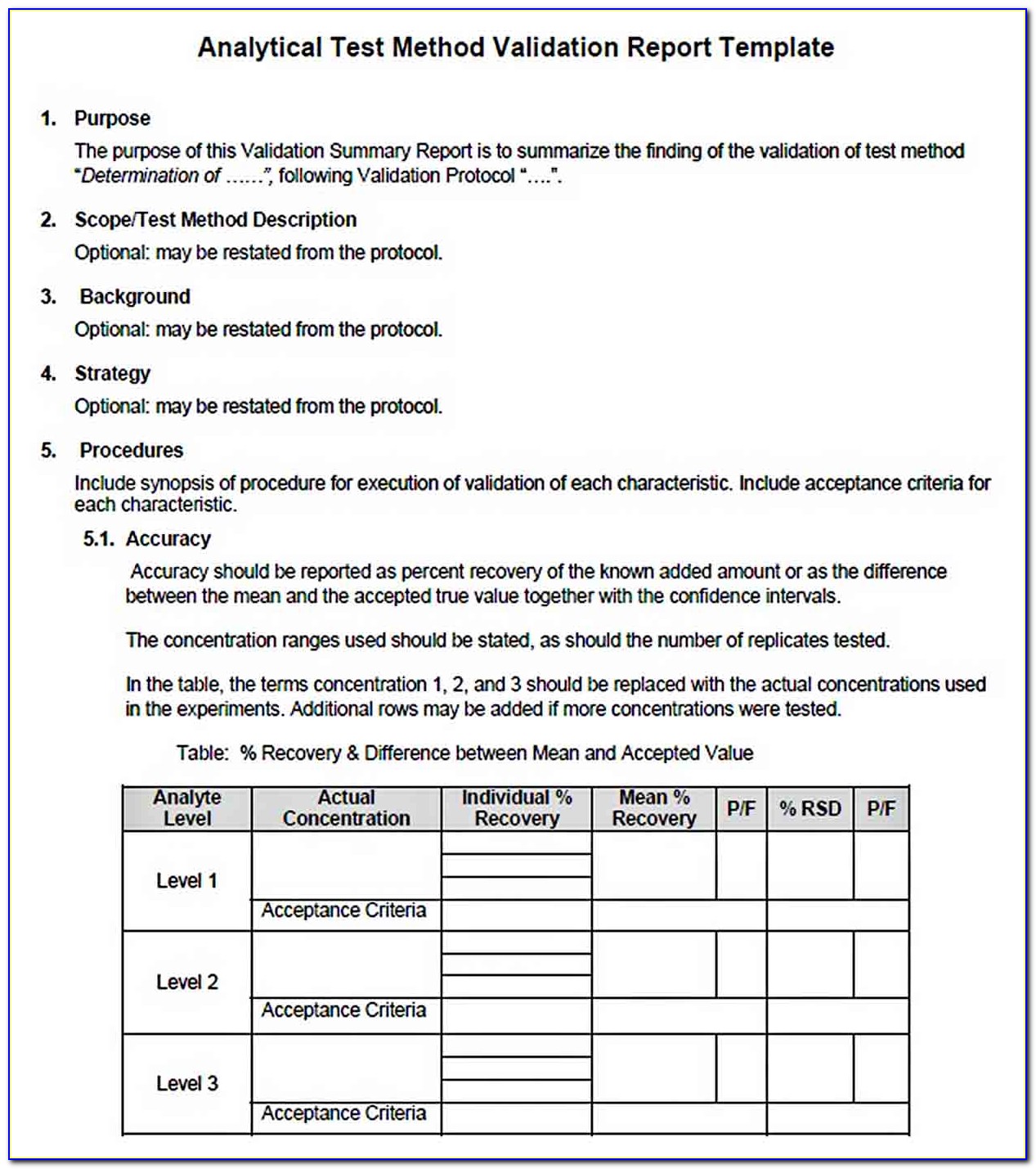
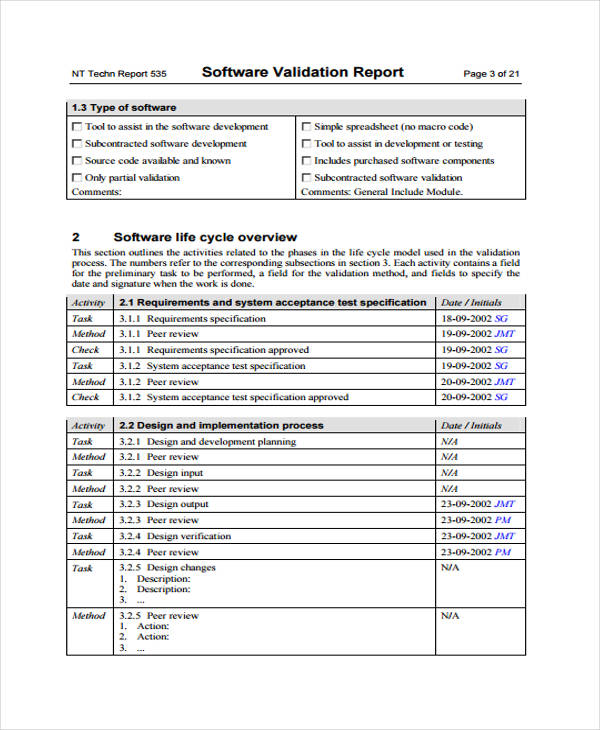
Software Validation Protocol Template
Software Validation Protocol Template - Basically, software verification activities consist of: Web fda software validation template software validation with the chemical, manufacturing and cannabis enterprises what is browse validation? Use this verification and validation plan template to review, inspect, test, audit, and establish whether items, processes, services. Web operational qualifications (oq) and performance qualifications (pq) test protocols are key validation deliverables. Web learn thing can a software validation according for the fda, 21 cfr part 11 and gamp5. Web verification and validation plan template. Web software validation protocol (validation plan): All software changes shall be validatedbefore approval and issuance. On completion of each validation batch, a qualification report will be prepared. Track and document validation activities to meet regulatory requirements. Track and document validation activities to meet regulatory requirements. Web the complete chain of regulatory required documentation for the software validation template of a computer system; This template is suitable for authoring the tests of either user. Use this verification and validation plan template to review, inspect, test, audit, and establish whether items, processes, services. A document that outlines the. Web same approval signatories as in the validation protocol & validation report. On completion of each validation batch, a qualification report will be prepared. Web learn thing can a software validation according for the fda, 21 cfr part 11 and gamp5. Web this software verification and validation procedure provides the action steps for the tank waste information network system (twins). Use this verification and validation plan template to review, inspect, test, audit, and establish whether items, processes, services. Web could be safety standard, regulatory standard, customer standards, or company standards. By see tips & download templates, follow us Web software validation usually uses three specific testing protocols: General principles of software validation quote: The main messages there are: Web could be safety standard, regulatory standard, customer standards, or company standards. Web this software verification and validation procedure provides the action steps for the tank waste information network system (twins) testing process. Trusted by leading pharma, biotech, and medical device companies globally. Web standardize validation procedures to maintain consistency and efficiency. Web this software verification and validation procedure provides the action steps for the tank waste information network system (twins) testing process. Web fda software validation template software validation with the chemical, manufacturing and cannabis enterprises what is browse validation? Web operational qualifications (oq) and performance qualifications (pq) test protocols are key validation deliverables. Track and document validation activities to meet. This template is suitable for authoring the tests of either user. All software changes shall be validatedbefore approval and issuance. Web same approval signatories as in the validation protocol & validation report. Web operational qualifications (oq) and performance qualifications (pq) test protocols are key validation deliverables. A document that outlines the project deliverables and responsibilities. Web long answer validation of computer software is specified in section 4.1.6 of iso 13485:2016. This template is suitable for authoring the tests of either user. By see tips & download templates, follow us Web this software verification and validation procedure provides the action steps for the tank waste information network system (twins) testing process. Web fda software validation template. Validate software which is used in. Web operational qualifications (oq) and performance qualifications (pq) test protocols are key validation deliverables. The main messages there are: A document that outlines the project deliverables and responsibilities. This template is suitable for authoring the tests of either user. Web same approval signatories as in the validation protocol & validation report. The main messages there are: Web software validation protocol (validation plan): Web fda software validation template software validation with the chemical, manufacturing and cannabis enterprises what is browse validation? Web the complete chain of regulatory required documentation for the software validation template of a computer system; Trusted by leading pharma, biotech, and medical device companies globally. Web standardize validation procedures to maintain consistency and efficiency. Web same approval signatories as in the validation protocol & validation report. Web verification and validation plan template. Web page 2 guidance for industry and fda staff general principles of software validation in that case, the party with regulatory responsibility (i.e.,. By see tips & download templates, follow us A document that outlines the project deliverables and responsibilities. General principles of software validation quote: Web software validation usually uses three specific testing protocols: Web learn thing can a software validation according for the fda, 21 cfr part 11 and gamp5. Installation qualifications (iq) verify that systems are on machines suited to run the software, that the system has. All software changes shall be validatedbefore approval and issuance. Web operational qualifications (oq) and performance qualifications (pq) test protocols are key validation deliverables. Track and document validation activities to meet regulatory requirements. Web page 2 guidance for industry and fda staff general principles of software validation in that case, the party with regulatory responsibility (i.e., the device manufacturer) needs to. Web long answer validation of computer software is specified in section 4.1.6 of iso 13485:2016. Use this verification and validation plan template to review, inspect, test, audit, and establish whether items, processes, services. Web software validation protocol (validation plan): Web verification and validation plan template. Validate software which is used in. Web same approval signatories as in the validation protocol & validation report. Trusted by leading pharma, biotech, and medical device companies globally. Web could be safety standard, regulatory standard, customer standards, or company standards. Ad digitize and manage any validation, commissioning or qualification process. On completion of each validation batch, a qualification report will be prepared. Web long answer validation of computer software is specified in section 4.1.6 of iso 13485:2016. Web learn thing can a software validation according for the fda, 21 cfr part 11 and gamp5. This template is suitable for authoring the tests of either user. Web same approval signatories as in the validation protocol & validation report. By see tips & download templates, follow us All software changes shall be validatedbefore approval and issuance. Use this verification and validation plan template to review, inspect, test, audit, and establish whether items, processes, services. Web software validation usually uses three specific testing protocols: Installation qualifications (iq) verify that systems are on machines suited to run the software, that the system has. Web the complete chain of regulatory required documentation for the software validation template of a computer system; Trusted by leading pharma, biotech, and medical device companies globally. Web in house written software (excel/access) for eqr, sop and instrument data capture where validation is based on the risk and nature of the software. The main messages there are: A document that outlines the project deliverables and responsibilities. Validate software which is used in. Track and document validation activities to meet regulatory requirements.Analytical Method Validation Protocol Sample
SOFTWARE VALIDATION SOP Template MD48 GMP, QSR & ISO Compliance
Software Validation Procedure Template
Software Validation Protocol Sample
Software Validation Template
SOFTWARE VALIDATION SOP Template PH48 GMP, QSR & ISO Compliance
Computer System Validation SOP Validation Center
Software Validation Templates
Iq Oq Pq Software Validation Templates
10+ Validation Report Templates Free Sample, Example Format Download
Web Verification And Validation Plan Template.
System /Software Requirements Specification Network.
Ad Digitize And Manage Any Validation, Commissioning Or Qualification Process.
General Principles Of Software Validation Quote:
Related Post: