Rt Template Health Canada
Rt Template Health Canada - Web sample forms & templates. Web as part of this project, on january 13, 2020, health canada announced that from april 1, 2020 to july 31, 2020, it will start accepting xml product monographs on a. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the. This page is intended to provide a number of sample forms and templates related to telehealth practice. Web dossier information 1 dossier type (required) pharmaceutical biologic company id (5 characters) (required) 2 company name (100 characters) (required) intended date of. Web health canada is responsible for helping canadians maintain and improve their health. To edit a queue specific template go to. If you are a consumer looking to report a problem with a medical device, access this online form. Sent via the cesg in folder 1.2.1 for. If you are an industry. Web as part of this project, on january 13, 2020, health canada announced that from april 1, 2020 to july 31, 2020, it will start accepting xml product monographs on a. If you are an industry. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the. Sent via the cesg. Numerous articles have shown that this variability exists across both quality and safety. The rep regulatory transaction (rt) and product information (pi) templates. Its purpose is to display the information as. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening in the province right now. This page is intended to provide a. Web as part of this project, on january 13, 2020, health canada announced that from april 1, 2020 to july 31, 2020, it will start accepting xml product monographs on a. Its purpose is to display the information as. This page is intended to provide a number of sample forms and templates related to telehealth practice. Web sample forms &. Recent work by sir brian. If you are an industry. Web you’ll need to indicate that you have a valid small business status on each fee form, application form or regulatory transaction (rt) template. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the. Web sample forms & templates. All the global templates are visible from this page. This page is intended to provide a number of sample forms and templates related to telehealth practice. Recent work by sir brian. Numerous articles have shown that this variability exists across both quality and safety. Its purpose is to display the information as. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening in the province right now. Web health canada is responsible for helping canadians maintain and improve their health. Sent via the cesg in folder 1.2.1 for. To edit a queue specific template go to. Its purpose is to display the information as. It should be noted that these forms and. Web sample forms & templates. Web health canada is responsible for helping canadians maintain and improve their health. This page is intended to provide a number of sample forms and templates related to telehealth practice. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening in the province right now. Numerous articles have shown that this variability exists across both quality and safety. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the. If you are a consumer. Web sample forms & templates. All the global templates are visible from this page. Web you’ll need to indicate that you have a valid small business status on each fee form, application form or regulatory transaction (rt) template. It should be noted that these forms and. Web there is a large amount of variability in health care today. The rep regulatory transaction (rt) and product information (pi) templates. If you are a consumer looking to report a problem with a medical device, access this online form. Device namefootnote 6 (required) device licence numberfootnote 7. All the global templates are visible from this page. Web rt, the effective reproductive number, reflects the current rate of transmission based on what. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening in the province right now. All the global templates are visible from this page. Web health canada is responsible for helping canadians maintain and improve their health. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the. This page is intended to provide a number of sample forms and templates related to telehealth practice. Its purpose is to display the information as. Device namefootnote 6 (required) device licence numberfootnote 7. Web you’ll need to indicate that you have a valid small business status on each fee form, application form or regulatory transaction (rt) template. Web as part of this project, on january 13, 2020, health canada announced that from april 1, 2020 to july 31, 2020, it will start accepting xml product monographs on a. Web sample forms & templates. To edit a queue specific template go to. It should be noted that these forms and. If you are a consumer looking to report a problem with a medical device, access this online form. Sent via the cesg in folder 1.2.1 for. The rep regulatory transaction (rt) and product information (pi) templates. Web dossier information 1 dossier type (required) pharmaceutical biologic company id (5 characters) (required) 2 company name (100 characters) (required) intended date of. Web there is a large amount of variability in health care today. If you are an industry. Numerous articles have shown that this variability exists across both quality and safety. Recent work by sir brian. Web you’ll need to indicate that you have a valid small business status on each fee form, application form or regulatory transaction (rt) template. The rep regulatory transaction (rt) and product information (pi) templates. Web there is a large amount of variability in health care today. Web as part of this project, on january 13, 2020, health canada announced that from april 1, 2020 to july 31, 2020, it will start accepting xml product monographs on a. All the global templates are visible from this page. Web dossier information 1 dossier type (required) pharmaceutical biologic company id (5 characters) (required) 2 company name (100 characters) (required) intended date of. It should be noted that these forms and. If you are an industry. Device namefootnote 6 (required) device licence numberfootnote 7. This page is intended to provide a number of sample forms and templates related to telehealth practice. Web health canada is responsible for helping canadians maintain and improve their health. To edit a queue specific template go to. Web rt, the effective reproductive number, reflects the current rate of transmission based on what is happening in the province right now. Recent work by sir brian. Numerous articles have shown that this variability exists across both quality and safety. Health canada requires the personal information to process regulatory application forms related to human and veterinary drug products under the.RT report sheet Respiratory therapist, Sheet, Etsy
Download Daily Route Plan Template for Free FormTemplate
COVID19 RT PCR Test • Corona Test Centre • Accredible • Certificates
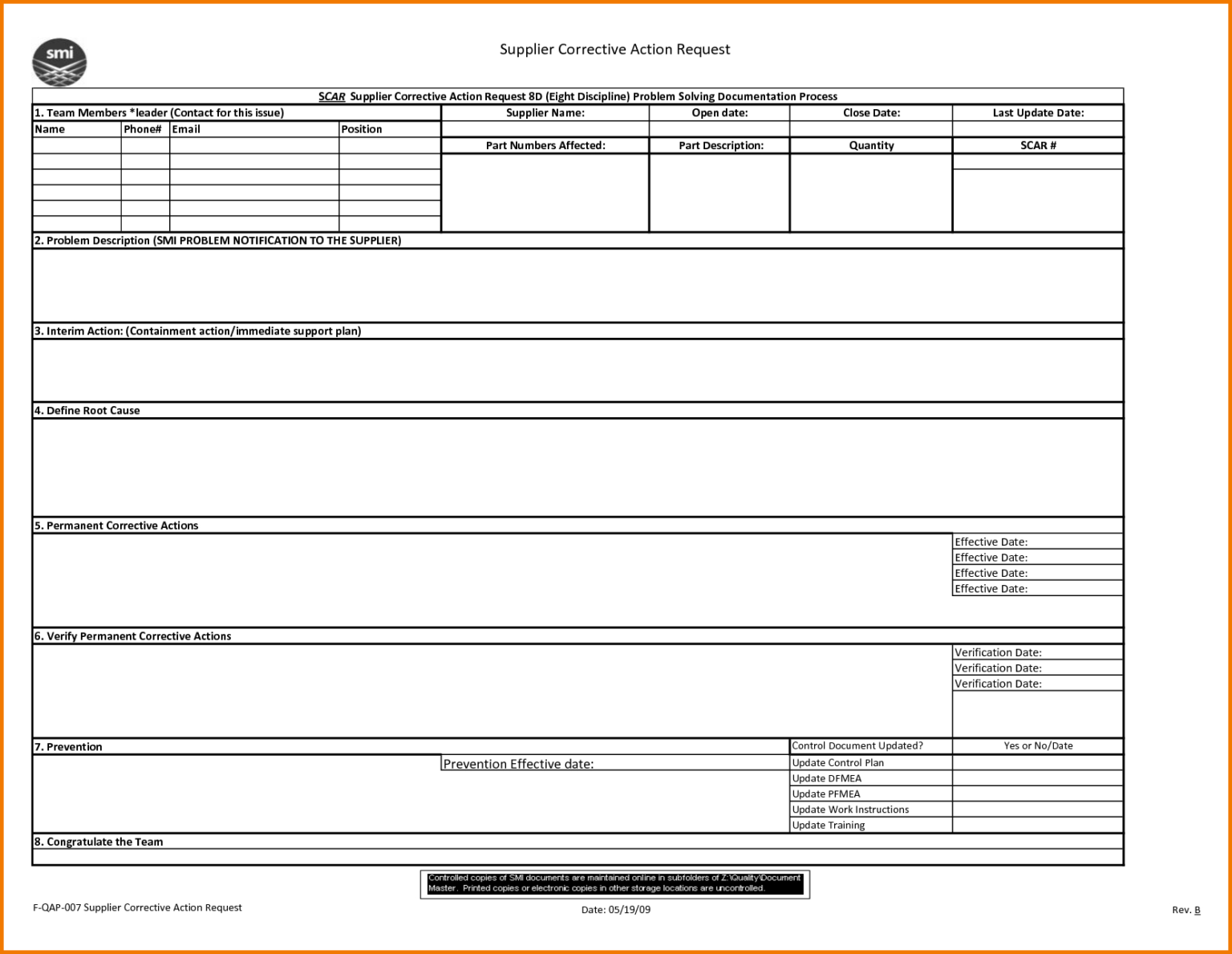
8D Report 2 Rt Template Pdf Excel Download Free Doc Examples pertaining
8D Report 2 Rt Template Pdf Excel Download Free Doc Examples with 8D
Canada Sample Medical Document for the Access to Cannabis for Medical
ATI Medication Template NUR1011 Studocu
Inspection Rt Template Home Build Your Own Version Top Excel with Home
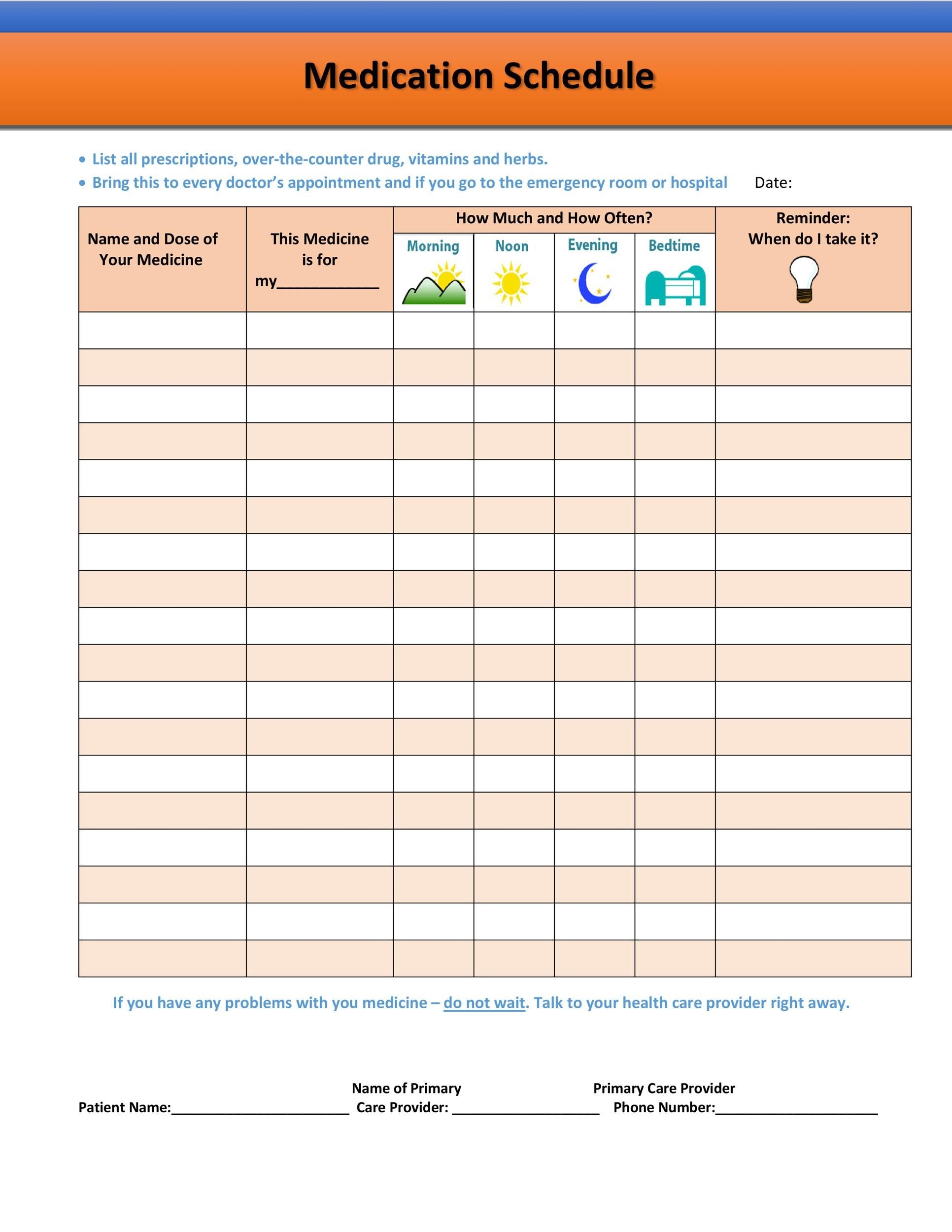
Free Printable Medication Chart Free Printable A to Z
Effective 28 Day Medication Calender Get Your Calendar Printable
If You Are A Consumer Looking To Report A Problem With A Medical Device, Access This Online Form.
Sent Via The Cesg In Folder 1.2.1 For.
Web Sample Forms & Templates.
Its Purpose Is To Display The Information As.
Related Post: