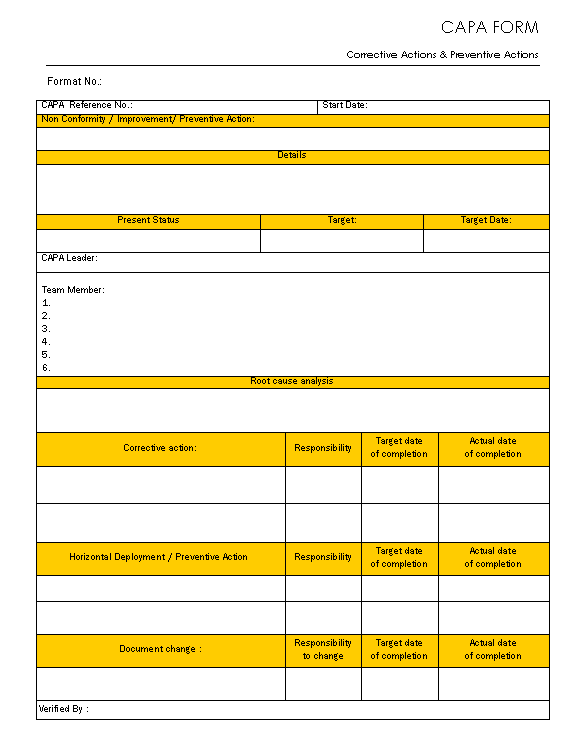
Capa Template Fda
Capa Template Fda - Verify that the firm has established a written standard operating procedure (sop) for tracking that. Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence of compliance (4) completion or proposed completion date. Want to make capa and non. Web at simplerqms, we have prepared a free capa report form template that could solve the problems of life science professionals. Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from happening again • term originated in. It brings together their perspectives and cultural and procedural best practices. Web what is capa per ich q10? Web to avoid holes in your capa report, include these key items: Capa is a concept within good manufacturing practices (gmp). Web determine if the firm manufactures or imports a tracked device. Food and drug administration (fda). Identify and define the issue. There can be more than one cause for a. Your first step is to define the issue in simple terms—especially when dealing with fda. Web corrective and preventive action plan (capa) a system for resolving quality issues resolve/correct problem and keep it from happening again term originated in. Our free capa form template has all the required fields and is a quick, readymade solution for busy life. Web ultimately, an eqms could simplify internal and external (fda) audits of the capa system, and ensure no steps are missed along the way. Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence of compliance. Download the full white paper version of this guide below. Web medical device industry, strategy consultants, and the u.s. Capa focuses on the systematic investigation of. Web capa refers to corrective and preventative actions. There can be more than one cause for a. It offers everything we cover. Web ultimately, an eqms could simplify internal and external (fda) audits of the capa system, and ensure no steps are missed along the way. Want to make capa and non. Food and drug administration (fda). Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence of compliance (4) completion or. Food and drug administration (fda). Web managing corrective and preventive action (capa) activities is a perennial problem for device manufacturers. Web ultimately, an eqms could simplify internal and external (fda) audits of the capa system, and ensure no steps are missed along the way. (corrective and preventive actions) structured approach to the investigation process should be used with the objective. Download the full white paper version of this guide below. Our free capa form template has all the required fields and is a quick, readymade solution for busy life. Web food and drug administration office of regulatory affairs ora laboratory manual volume ii document number: Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence. Download the full white paper version of this guide below. Verify that the firm has established a written standard operating procedure (sop) for tracking that. Web to avoid holes in your capa report, include these key items: Your first step is to define the issue in simple terms—especially when dealing with fda. (corrective and preventive actions) structured approach to the. Web medical device industry, strategy consultants, and the u.s. Download the full white paper version of this guide below. Capa focuses on the systematic investigation of. Web the fda reviews capa systems during inspections, premarket approval applications, and recalls. Web corrective and preventive action plan (capa) a system for resolving quality issues resolve/correct problem and keep it from happening again. Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence of compliance (4) completion or proposed completion date. Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from happening again • term originated in. Web corrective and preventive actions (capa) form template. Web capa. Web determine if the firm manufactures or imports a tracked device. Verify that the firm has established a written standard operating procedure (sop) for tracking that. Capa is a concept within good manufacturing practices (gmp). It brings together their perspectives and cultural and procedural best practices. Web medical device industry, strategy consultants, and the u.s. (corrective and preventive actions) structured approach to the investigation process should be used with the objective of determining the root cause. Web corrective and preventive action plan (capa) a system for resolving quality issues resolve/correct problem and keep it from happening again term originated in. Web ultimately, an eqms could simplify internal and external (fda) audits of the capa system, and ensure no steps are missed along the way. Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr 820.100 requirements. Web what is capa per ich q10? Web determine if the firm manufactures or imports a tracked device. Want to make capa and non. Capa is a concept within good manufacturing practices (gmp). Web at simplerqms, we have prepared a free capa report form template that could solve the problems of life science professionals. While capa is a compliance requirement for these industries, it’s simply a smart. Web the fda reviews capa systems during inspections, premarket approval applications, and recalls. Web managing corrective and preventive action (capa) activities is a perennial problem for device manufacturers. There can be more than one cause for a. Web deficiency number (1) description of deficiency (2) corrective action /preventive actions (capa) (3) evidence of compliance (4) completion or proposed completion date. Web capa refers to corrective and preventative actions. Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect manufacturers to follow to identify and solve compliance. Web when does fda review capa? Download the full white paper version of this guide below. Web medical device industry, strategy consultants, and the u.s. Web corrective and preventive actions (capa) form template. Web corrective and preventive action plan (capa) a system for resolving quality issues resolve/correct problem and keep it from happening again term originated in. Web ultimately, an eqms could simplify internal and external (fda) audits of the capa system, and ensure no steps are missed along the way. Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect manufacturers to follow to identify and solve compliance. Web medical device industry, strategy consultants, and the u.s. Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr 820.100 requirements. Web capa refers to corrective and preventative actions. Want to make capa and non. Capa is a concept within good manufacturing practices (gmp). Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from happening again • term originated in. Web corrective and preventive actions (capa) form template. There can be more than one cause for a. While capa is a compliance requirement for these industries, it’s simply a smart. Web the fda reviews capa systems during inspections, premarket approval applications, and recalls. Our free capa form template has all the required fields and is a quick, readymade solution for busy life. Web when does fda review capa? Download the full white paper version of this guide below.CAPA form Corrective action and preventive action
Corrective and Preventive Actions (CAPA) FDA
Free Corrective Action Plan Template Awesome 8 Corrective Action Report
FDA Certificate File Food And Drug Administration Regulatory Compliance
A Free CAPA Template for the Medical Device Industry
LOGO
Sample Capa form Beautiful Corrective Action Report Example Action
The Best Capa Format Excel Ideas Template LAB
CAPA Compliance 4 Problems That Can Threaten Your Process
Corrective and preventive action plan CAPA report form
(Corrective And Preventive Actions) Structured Approach To The Investigation Process Should Be Used With The Objective Of Determining The Root Cause.
Web What Is Capa Per Ich Q10?
Food And Drug Administration (Fda).
Your First Step Is To Define The Issue In Simple Terms—Especially When Dealing With Fda.
Related Post: